Persistent or Suspected-Resistant Hookworm Infections
Pablo David Jimenez Castro, DVM, University of Georgia
Ray M. Kaplan, DVM, PhD, DEVPC, DACVM (Parasitology), St. George's University, Grenada, West Indies
Numerous cases of canine hookworm (ie, Ancylostoma caninum) with multidrug resistance to all 3 major anthelmintic classes have been identified.1
Background & Pathophysiology
Diagnostic surveillance performed at the authors’ laboratory over the past few years suggests the presence of multidrug-resistant (MDR) hookworms (ie, A caninum) likely evolved on greyhound breeding farms and in racing kennels. Most, if not all, actively racing and/or recently adopted greyhounds appear to be infected with MDR hookworms; however, many cases of MDR hookworms have been diagnosed in non-greyhound breeds, suggesting MDR hookworms are spreading to the general canine population.
Hookworms (Figure 1) have a direct life cycle, with adult females releasing a large number of eggs (up to 10,000/day). Once passed in the feces, development of eggs to third-stage infective larvae (L3) typically takes ≈5 days, although this will vary depending on temperature. Dogs may be infected via both the oral and percutaneous routes. L3 larvae are ingested either directly or by ingestion of paratenic hosts carrying L3 tissue larvae. After penetrating the skin, L3 larvae migrate via the bloodstream to the lungs, penetrate the alveoli, migrate up the bronchial tree to the trachea, are expectorated via coughing, are swallowed, and enter the small intestine, where they complete development into the adult stage. The prepatent period for either route of infection is 15 to 26 days. Following skin penetration in dogs older than 3 months of age, A caninum L3 larvae often undergo somatic migration to the muscle, fat, and other organs; encyst; and enter a hypobiotic state.
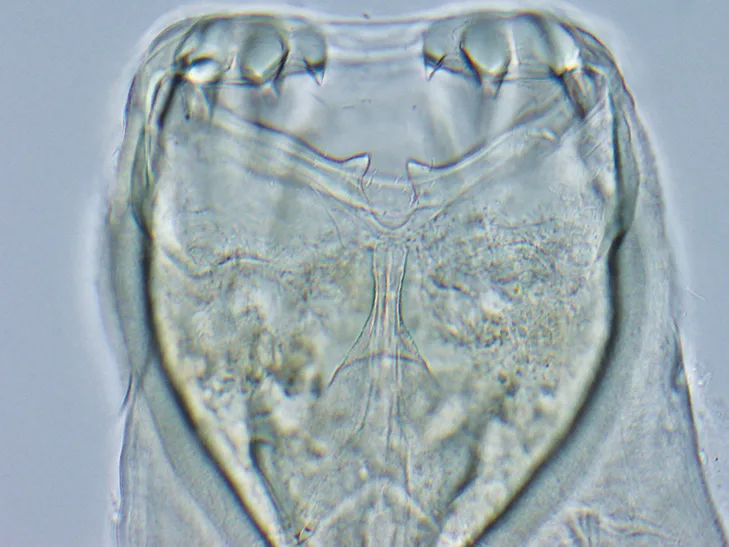
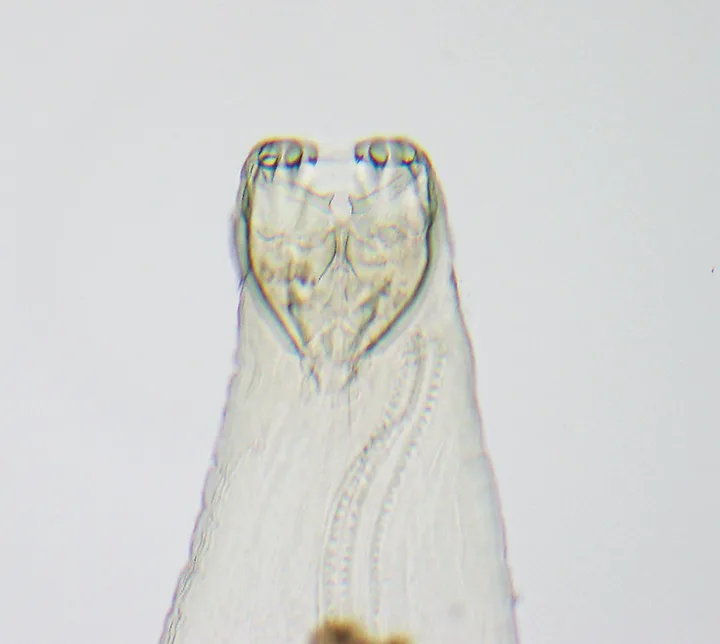
Anterior end of adult A caninum. The buccal capsule (ie, mouth) contains the characteristic 3 pairs of teeth.
Encysted somatic larvae may become reactivated under 2 conditions: host pregnancy or larval leakage (ie, when arrested somatic larvae continuously leak from tissue and complete migration to the intestine, where they develop into adults and begin a new round of egg shedding). An important mode of A caninum transmission is the transmammary route, in which puppies become infected by reactivated larvae that migrate to the mammary tissue of the dam.
Although direct evidence is lacking, based on the authors’ observations and previous research on this issue in the sheep parasite, Haemonchus contortus,2 it is probable that macrocyclic-lactone resistance has worsened as the use of moxidectin has become more common. Further, drug resistance in nematodes is typically a slow evolutionary process, requiring many years of drug selection to reach levels that are clinically apparent.3 This was most likely the case for A caninum; thus, the level of resistance seen in any particular case and to any particular drug will depend in part on the time frame of the animal’s adoption and previous anthelmintic treatments.
The emergence and spread of MDR hookworms that are poorly responsive to typical anthelmintic treatments necessitate a different management approach.
Diagnosis
When addressing persistent cases of A caninum infection, the clinician should first differentiate between larval leakage4 with drug-susceptible A caninum and infection with MDR hookworms.
There are 3 methods to diagnose anthelmintic resistance: performing a fecal egg count reduction test (FECRT), submitting a sample to a laboratory that can perform in vitro drug bioassays with hookworms, and submitting a sample to a laboratory that can perform molecular testing for resistance.
FECRT is the ideal practical approach, as laboratory expertise and facilities may not be readily available for the other diagnostic methods. FECRT can be easily accomplished at the clinic level for minimal cost.
Performing a Fecal Egg Count Reduction Test
To perform an FECRT, the number of eggs per gram of feces must be quantified pre- and posttreatment. Fecal flotations, which are frequently performed in small animal practice, are inadequate for an FECRT. The pretreatment fecal sample can be collected either the day before or the day of treatment and should be kept refrigerated until submission to the laboratory to prevent development and hatching of eggs prior to testing.
To evaluate for resistance, a quantitative fecal egg count (FEC) method (eg, McMaster,5 Mini-FLOTAC6; Figures 2 and 3) is needed. A double-centrifugation method (eg, modified Wisconsin [Figure 4]) could also be used but is more time consuming, more labor intensive, less accurate, and less precise.7,8 A quantitative method is necessary to assess response to treatment. This approach is standard for diagnosis of anthelmintic resistance in livestock nematode parasites, for which drug resistance has been a long-standing problem. Further, because only 1 dog is typically being tested, as compared with groups of 10 to 20 livestock, the authors also recommend performing 2 separate FECs on the pretreatment sample and 2 separate FECs on the posttreatment sample. The FEC reduction is then calculated by comparing the average eggs per gram (EPG) for the 2 pretreatment FECs and the average EPG for the 2 posttreatment FECs. By repeating the FEC, the variability of each FEC measurement will be reduced by half, thus improving the accuracy of the measured FEC reduction.
A specialized laboratory that offers the service should be contacted if in-clinic or diagnostic laboratory FEC testing is not an option. McMaster slides, Mini-FLOTAC reading discs, and Fill-FLOTAC devices are available for purchase (see Fecal Egg Count Reduction Test Resources).

FIGURE 2
McMaster slide
Fecal Egg Count Reduction Test Resources
McMaster Slides
Mini-FLOTAC Devices
University of Georgia is serving as the North American distributor for University of Naples, the manufacturer of the Mini-FLOTAC system. To place an order:
Email famacha@uga.edu
Visit the University of Naples website: parassitologia.unina.it/flotac/mini-flotac/?lang=en
Posttreatment Fecal Egg Count
During testing, the most-recently used anthelmintic should be readministered, even if it elicited poor therapeutic results. Use of an alternate drug will not allow differentiation between larval leakage and resistance as the cause of treatment failure, as the worms may not be resistant to the new drug. However, this may only be a theoretical concern, as the authors’ experience indicates that drug-resistant hookworm infrapopulations most likely will be MDR to all 3 anthelmintic classes.
Multiple days (≥3) are needed for eggs already shed in the intestine to be fully cleared.9 In addition, the authors have observed a temporary, but high, level of suppression on worm fecundity following fenbendazole treatment. A 99% reduction in FEC has been observed by 3 days posttreatment, with egg counts rapidly rising again after ≈10 days.1,10 This phenomenon has been reported rarely in sheep after treatment with benzimidazole anthelmintics11 and on multiple occasions in strongylids of ruminants12-14 and pigs15 following treatment with ivermectin and moxidectin. Consequently, checking FEC too soon posttreatment can yield a false-negative result for resistance.
In contrast, if too much time passes, larvae arrested in somatic tissue could repopulate the intestine and begin a new round of egg shedding, leading to a false-positive result. The prepatent period for A caninum has been reported to be as early as 14 days,16-18 but this time frame is from studies in immune-naive puppies following primary infection. Few data exist on time to worm maturity and egg production in older dogs with chronic infections; however, based on clinical data and other reports,19 new worms take 3 to 4 weeks to repopulate the lumen of the small intestine and initiate a new round of egg shedding.
The following timeframes are thus recommended for posttreatment FEC: 10 to 14 days after treatment with pyrantel, 14 days after treatment with fenbendazole/febantel, and 14 days after treatment with moxidectin.
The following formula can be used to calculate FECRT percentage:

Interpretation of Fecal Egg Count Reduction Test Results
FECRT results should be interpreted conservatively, as FEC can be highly variable. It is important to note, however, that when commonly used anthelmintics were first approved, high efficacies were reported based on worm counts (>99% for febantel, moxidectin, and milbemycin oxime20-22; >98% for fenbendazole23; variable for pyrantel, with a mean across studies of ≈94% and over half of studies yielding >99%24).
In contrast, in a recent study using an MDR A caninum isolate (Worthy 4.1F3P), the efficacies based on worm counts were 23%, 9%, and 26% for pyrantel, milbemycin oxime, and fenbendazole, respectively.10 The corresponding FEC reductions measured 10 days posttreatment for these same treatments were 13%, 0%, and 46%, respectively.10 These data demonstrate that poor FEC reduction can be expected against an MDR A caninum isolate following treatment with typical commercial products.
The following interpretation of FEC reduction results are suggested:
<75% reduction: indicative of resistance (larval leakage is highly unlikely to be the cause of persistent egg shedding)
75% to 89% reduction: suggestive of resistance (larval leakage is unlikely to be the cause of persistent egg shedding)
90% to 95% reduction: suggestive of reduced efficacy and should raise suspicion for resistance, but results should be viewed as inconclusive (persistent egg shedding could be due to resistance or larval leakage)
>95% reduction: suggestive of effective treatment (larval leakage is likely the cause of persistent egg shedding)
FECRT is sensitive for detecting resistance (ie, dead worms do not shed eggs); consequently, effective treatment will produce a high reduction in the number of eggs shed, and a poorly effective treatment will yield a low reduction in eggs shed. However, egg-shedding levels on a per-worm basis can vary greatly, and egg production per worm can increase following treatment that kills some of the worms (referred to as density dependent fecundity).25 Therefore, the actual percentage for reduction should not be overinterpreted; for example, 25% and 70% reduction both indicate resistance, but the results should not be interpreted as being greatly different. Likewise, given the expected variability, it should not be assumed that the reduction in FEC will be the same in each case of resistance or even in the same dog if the FECRT is repeated.
FEC reduction between 75% and 95% yields an inconclusive result; repeating FECRT at the next treatment is advised.
Definitive Diagnosis
A diagnosis of resistance should only be established if all of the following are true:
The patient was treated with the proper dosage.
The drug administered was within the expiration date and stored properly.
Fecal samples were labeled and stored correctly prior to fecal analysis.
An FECRT was performed.
Proper laboratory techniques were applied when conducting the FECRT, and the same method was used on both the pre- and posttreatment samples.
Treatment & Follow-Up
The treatment plan should depend on the results of the FECRT. If FEC reduction is >95%, treatment should be considered effective. Drugs are not 100% effective, even against drug-susceptible worms; thus, some eggs may be seen, particularly when pretreatment FEC is high. Because resistance can be ruled out, eggs seen on previous fecal examinations are most likely a result of larval leakage. The patient should be treated with an anthelmintic monthly, and fecal examinations should be conducted every few months. Moxidectin can be a good choice in dogs with larval leakage, although any effective anthelmintic should work.
If FEC reduction is between 90% and 95%, FECRT should be repeated a few weeks later at the next treatment.
If FEC reduction is between 75% and 90%, FECRT can be repeated for more conclusive results, or because there is a high chance the worms are resistant, the treatment plan suggested below can be followed.
If FEC reduction is <75%, treatment should be considered ineffective and adjusted to a triple anthelmintic combination with all drugs administered concurrently at the labeled doses. Drugs should be administered sequentially on the same day and not mixed together.
This treatment plan has been successful in eliminating active infections in persistent hookworm cases26:
Febantel (25 mg/kg PO)/pyrantel pamoate (5 mg/kg PO)/praziquantel (5 mg/kg PO) + moxidectin (2.5 mg/kg topical), or
Fenbendazole (50 mg/kg PO once daily for 3 days) + pyrantel pamoate (5 mg/kg PO) + moxidectin (2.5 mg/kg topical)
Treatment success using this triple-drug combination depends on whether the hookworms are moxidectin-resistant. MDR hookworms studied by the authors were all ivermectin-resistant but may still be moxidectin-sensitive. The aforementioned regimen should be effective if hookworms are moxidectin-sensitive; however, this approach may be ineffective if the infecting source hookworms were previously treated with moxidectin. The authors have diagnosed recent cases of moxidectin-resistant A caninum in greyhounds in which monthly moxidectin treatments offered little benefit. If this monthly treatment regimen is effective in eliminating egg shedding, the patient will need to remain on this treatment for several months, or possibly for life, as somatic tissue stores will continually leak and repopulate the intestine for an extended time.
Extra-label administration at higher-than-label doses might improve efficacy, but there are currently no data to support such a recommendation. In addition, some parasitologists recommend repeating moxidectin treatment every 2 weeks for the first 4 treatments, then treating monthly, as this allows the moxidectin to rapidly reach a steady-state tissue concentration due to the long half-life of moxidectin in dogs.27-29 This is reasonable and potentially beneficial, although no specific data presently exist. Other products containing moxidectin may also be effective, but there are no published data to support the effectiveness of those products against MDR A caninum isolates.
Considerations for Extra-Label Emodepside
Due to limited available data, the authors do not recommend extra-label emodepside treatment; however, clinicians can use this information to evaluate whether use might be appropriate.
Emodepside topical solution for cats has no efficacy in dogs when administered topically; it must be administered orally.
Drug formulation matters, and each pharmaceutical product is carefully formulated to optimize pharmacokinetics and drug safety; the excipients used for topical products are not intended for oral use and may lead to variability in the pharmacokinetic and safety profiles.
The suggested dose of emodepside for dogs with MDR A caninum isolates is 1 mg/kg PO.
Emodepside is a known substrate for P-glycoprotein,32 and dogs with a deletion mutation of the multidrug sensitivity gene (MDR1 gene, also known as ABCB1 gene) may be at increased risk for severe adverse effects,33 especially if they receive incorrect doses of the topical feline product.
The 1 mg/kg PO dose of emodepside for dogs is one-third of the topical labeled dose for cats, and administering more than this dose may increase the likelihood of adverse effects.
Given the potential risks of using this product in dogs, emodepside topical solution for cats should only be used in dogs when the poor effectiveness of the triple anthelmintic combination has been previously confirmed via FECRT.
All FDA requirements and Animal Medicinal Drug Use Clarification Act provisions with regard to extra-label drug use should be closely followed, including informing owners about label warnings and other known risks.
Emodepside
If the triple combination approach is ineffective, emodepside is the only potentially effective alternative treatment, based on a recent study evaluating the efficacy of emodepside and praziquantel against an MDR A caninum isolate.10 Oral emodepside (1 mg/kg) with praziquantel (5 mg/kg) demonstrated an efficacy of 99.6% with a 100% reduction in FEC at 10 days posttreatment.
Emodepside is not currently approved for use in dogs in the United States; however, emodepside (with praziquantel) is FDA-approved as a topical solution for cats. The authors have determined that extra-label use of emodepside topical solution for cats, administered PO at a different dose than is recommended for this product, has high efficacy in MDR A caninum isolates refractory to triple combination treatment.
The authors have monitored FEC in 17 client-owned dogs, both greyhounds and nongreyhounds, treated by private practice clinicians using 1 mg/kg PO emodepside administered once. In all cases, FEC reduction was 100% at 14 days, and no adverse effects were observed. However, there are a number of important factors to consider with extra-label emodepside use (see Considerations for Extra-Label Emodepside).
If extra-label use of the emodepside topical solution for cats is warranted, the patient should be fasted overnight prior to administration, and food should not be provided until 4 hours posttreatment. The dose should be given at the clinic and not dispensed to the owner. The product should be drawn into a syringe with a needle; then, the needle should be removed and the syringe administered as distal orally as possible to decrease the ability of the patient to taste the product.
Precise dosing of emodepside is critical and cannot be readily achieved without careful calculations. The correct canine dose cannot be estimated based on the feline label. The product comes in 3 sizes: small cat (5.5 lb [2.5 kg]), medium cat (11 lb [5 kg]), and large cat (17.6 lb [8 kg]), all of which have a different volume but the same concentration of emodepside (21.4 mg/mL). The following formula should be used to determine the correct dose for dogs:

For example, an 8.8-lb (4-kg) dog would receive 0.19 mL, and a 66-lb (30-kg) dog would receive 1.4 mL.
Other Supportive Treatments
The authors have not evaluated nor are aware of any evidence regarding the use of other concurrent and/or supportive treatments (eg, probiotics) and cannot provide recommendations on their use.
Follow-Up & Environmental Hygiene
The authors strongly recommend that FEC (not just flotations) be evaluated monthly to monitor egg shedding.
It is critical that strict environmental hygiene is practiced. Feces of a dog shedding hookworm eggs (or any helminth parasite) should be picked up immediately and properly disposed of to eliminate the potential for reinfection or spread.
It takes ≥5 days for hookworm eggs to develop to the infective third-stage larvae in ideal temperature and humidity conditions30; therefore, fecal pickup even every few days can be highly effective in preventing environmental contamination. However, waiting can result in feces breakdown, allowing the hookworm larvae (or other parasite eggs/larvae) to contaminate the environment.
If reinfection with ivermectin-resistant worms from the environment is permitted to occur in dogs treated with moxidectin, resistance to moxidectin can rapidly develop.31
There are several methods for killing hookworm larvae in the environment, but their effectiveness is undetermined.
The recommendations made in this article are based on the authors’ interpretation of best available evidence at the time of publication and should not be construed as being permanent. As new knowledge is gained and new products become available, recommendations listed here are likely to change.
EPG = eggs per gram, FEC = fecal egg count, FECRT = fecal egg count reduction test, L3 = third-stage infective larvae, MDR = multidrug-resistant