Isoxazolines
Craig Datz, DVM, MS, DABVP (Canine & Feline), DACVN, University of Missouri
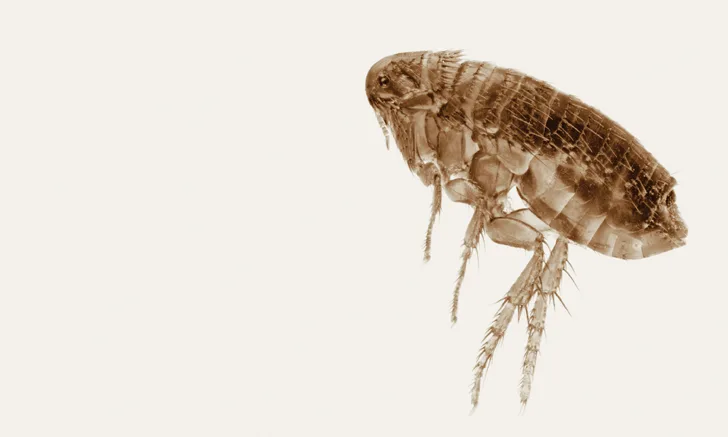
Isoxazolines kill fleas and are indicated for the treatment and prevention of flea infestations, as well as the treatment and control of various tick infestations.1-6
Overview
The isoxazoline drug class was launched in the United States in 2014 with afoxolaner, followed shortly by oral fluralaner, sarolaner, topical fluralaner, and lotilaner.
Mechanism of Action
Isoxazolines are absorbed systemically; fleas and ticks must bite the animal to be killed.
Isoxazolines work by selective inhibition of GABA- and glutamate-gated chloride channels, leading to hyper-excitation and death of the flea or tick.7-10
Because GABA channels in mammals have much a lower sensitivity to isoxazolines and mammals lack anion-inhibitory glutamate channels, there is low toxicity potential.8
For fleas, the onset of action for all products is reported to be 2 to 4 hours, with nearly 100% of fleas killed within 8 hours1-6; for ticks, the onset of action for >90% tick control is 4 to 8 hours, although study protocols often assess tick control at 48 hours after administration.1-6
Clinical Application
Several studies have shown that isoxazolines can reduce the risk for tick-borne disease transmission.
Afoxolaner and sarolaner each prevented Borrelia burgdorferi infection (ie, Lyme disease) in controlled laboratory studies.11,12
In other laboratory studies, afoxolaner, fluralaner, and lotilaner each prevented transmission of Babesia canis.13-16
Ixodes holocyclus, the Australian paralysis tick, was controlled by afoxolaner, fluralaner, and sarolaner.17,18
In a comparative laboratory study, transmission of Ehrlichia canis was prevented by permethrin–imidacloprid and prevented in some but not all dogs by either afoxolaner or fluralaner.19
Administration
Fluralaner and lotilaner chewables should be administered with food, whereas afoxolaner and sarolaner may be given with or without food.1-3,6
Labeled ages, body weights, and dosing intervals vary (see Table).
Collies with the multidrug sensitivity gene (MDR1 gene, also known as ABCB1 gene) mutation (ivermectin-sensitive) were treated with up to 10 times the label dose of afoxolaner or 3 times the label dose of fluralaner with no adverse effects noted.20,21
Fluralaner has been shown to be well tolerated with concurrent use of milbemycin–praziquantel and deltamethrin collars in dogs and emodepsid–praziquantel in cats.22-24
Extra-label use of isoxazolines for other ectoparasites has been reported.
In one study of generalized demodicosis in 8 adult dogs, a single dose of fluralaner resulted in elimination of Demodex canis mites and resolution of dermatologic signs.25
Afoxolaner, sarolaner, and lotilaner at label doses were also shown to be effective in dogs with generalized demodicosis.26-29
In dogs with Sarcoptes scabiei var canis, 2 doses of afoxolaner (on days 0 and 28) or sarolaner (on days 0 and 30) or a single dose of fluralaner eliminated mites and resulted in skin improvement within 4 weeks.30-33
Dogs with ear mites (Otodectes cynotis) have been successfully treated with afoxolaner, fluralaner, and sarolaner; topical fluralaner is effective in cats with ear mites.28,34,35
Table 1. Isoxazolines at a glance1-5
*The administration interval is every 8 weeks for Amblyomma americanum (lone star) ticks.2,4
Safety & Adverse Effects
Vomiting, diarrhea, lethargy, and decreased appetite were occasionally observed in safety studies in puppies 8 to 9 weeks old (in both treated and control groups) at 1 time, 3 times, and 5 times the maximum oral label dose and in field studies.1-4,6,9,36-38
Neurologic signs (eg, tremors, seizures) were noted in some puppies treated with sarolaner at 3 to 5 times the label dose.3
Fluralaner topical solution was similarly studied in dogs and cats; other than cosmetic changes at the application sites, no treatment-related adverse effects were observed.4,5
In the feline field study, neurologic signs were seen with topical fluralaner in 2 of 224 cats5; this drug should be used with caution in cats with a history of neurologic disease.5
Safety in breeding, pregnant, or lactating dogs has not been evaluated for afoxolaner, sarolaner, or lotilaner.1,3,6
Oral fluralaner was studied at up to 3 times the maximum label dose at 8-week intervals in male and female beagles through breeding, pregnancy, and lactation.
No treatment-related effects were observed in the adult dogs or on reproductive performance.
In litters from 2 of 10 dams, abnormalities (eg, limb deformity, cleft palate) were noted in some puppies on gross examination.2
In uncontrolled, open-label field studies, all drugs were effective in reducing or resolving signs of flea allergy dermatitis in dogs.39-43