Canine Melanoma Vaccine
Heather Wilson-Robles, DVM, DACVIM (Oncology), Texas A&M University
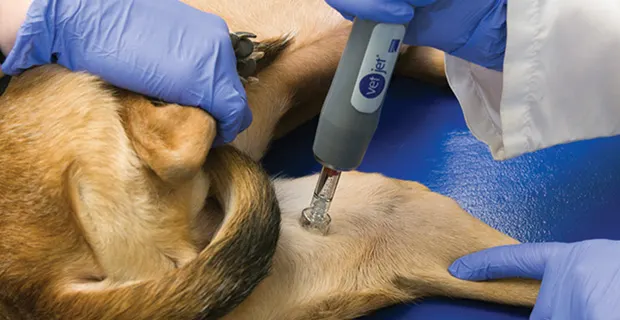
Administration of melanoma vaccine in the area of the medial thigh
Malignant melanoma is the most common malignant oral tumor in dogs, but it can also occur in other locations (eg, nail bed, eye, perineum, nasal cavity, GI tract, skin).1,2
Malignant melanomas most often occur in older dogs and in the Scottish terrier, golden retriever, poodle, and dachshund breeds2; poodles also may be at higher risk for developing malignant melanocytic tumors.1 The metastatic rate can vary according to physical location. Metastasis has been reported in 60% of oral melanomas, 40% of digital melanomas, and 12% of cutaneous melanomas.3

Oral melanoma in a canine patient
Indications
Oncept (Merial, petcancervaccine.com), immunotherapy labeled for dogs with stage II or III oral melanoma, can help achieve local disease control.4 This therapy uses an orthologous DNA (different species) vaccine against tyrosinase, a melanosomal glycoprotein needed for melanin synthesis, to induce an antityrosinase immune response.5 Tyrosinase has been over-expressed in melanomas of various species.6-8 Although the human tyrosinase protein differs enough from the canine tyrosinase protein to allow human tyrosinase protein stimulation of an immune response in canine patients, in some cases the human tyrosinase protein is similar enough to the canine tyrosinase protein that the immune response is effective against canine melanoma cells.4
This vaccine works best when the primary tumor has been removed and there is no evidence of regional or distant metastasis (or metastases have been removed). The vaccine may also be useful after radiation therapy for tumors that are not surgically excisable. The vaccine is used for dogs with gross disease, although objective evidence of response in this setting is lacking.9
Traditional World Health Organization TNM-Based Staging Scheme for Dogs with Oral Melanoma10
Stage I = T1, N0, M0; Stage II = T2, N0, M0; Stage III = T2, N1, M0 or T3, N0, M0; Stage IV = any T, any N, M1
Contraindications
According to the vaccine’s label, there are no contraindications; however, use of the vaccine is not likely to be successful in a gross disease setting, although anecdotal responses have been noted.
In rare instances, the vaccine has reportedly caused lethargy, fever, and inflammatory or hypersensitivity reactions.4 The most common adverse effects are redness or swelling and mild pain at the vaccination site.
Advantages
The strongest advantage of this therapy is its ability to systemically treat malignant melanoma with minimal to no adverse effects. Chemotherapy, another systemic option, can have significant toxicity in patients.

Melanoma vaccine (Oncept, petcancervaccine.com)
In addition, chemotherapy has not been shown to be particularly efficacious in dogs, with the highest overall response rates being reported for carboplatin. These response rates are generally less than 30% and often lead to only a modest increase in survival time.3,4 Local therapies, such as surgery or radiation, lack the systemic benefits of the vaccine or chemotherapy.
Disadvantages
The primary disadvantages to this vaccine are price and limited availability. The average cost per injection is $$$ to $$$$ (see Cost Key), making the overall cost comparable with that for chemotherapy. Dogs receive an initial induction series of 4 injections 2 weeks apart and then a booster q6mo. Currently, the vaccine is only available to veterinary oncologists (or internists in regions without oncologists).
Clinical Impact
The vaccine has been widely used in the United States for several years. When used in conjunction with local control, it appears to be well tolerated, safe, and efficacious for dogs with stage II (and possibly stage III) disease. The advent of commercially available immunotherapy for dogs with cancer is exciting and will hopefully spur similar therapeutics.
A Case Study5
Oncept appeared to significantly improve survival times in a study comparing dogs with stage II or III malignant melanomas undergoing local disease control (n = 58) with historical control dogs treated with surgery alone (n = 53).
Stage II
Because <50% of vaccinated dogs died before observation ended, median survival time (MST) of vaccinated dogs was not determined.
MST for control dogs was 324 days (p<0.001).
Stage III
Patients (n = 14 vaccinated, 19 control dogs) were not well delineated by treatment.
MST of vaccinated dogs was 235 days.
MST = median survival time